Table of Contents
Validity under §112
Supreme Court un-defines "definiteness" with "reasonable certainty" (updated 08/05/15)
 In Nautilus v. Biosig Instruments (cached), the Supreme Court vacated and remanded to the Federal Circuit its reversal and remand of the district court's conclusion that that the phrase “spaced relationship” of electrodes of a heart rate monitor was indefinite. (If the electrodes touched, then that would short the circuit, and the heart rate of a person holding the sensor could not be determined.) The Supreme Court specifically overturns Federal Circuit precedent stating that, a claim is indefinite “only when it is 'not amenable to construction' or 'insolubly ambiguous'” 1). Instead, the Supreme Court holds that § 112 ¶2 requires:
In Nautilus v. Biosig Instruments (cached), the Supreme Court vacated and remanded to the Federal Circuit its reversal and remand of the district court's conclusion that that the phrase “spaced relationship” of electrodes of a heart rate monitor was indefinite. (If the electrodes touched, then that would short the circuit, and the heart rate of a person holding the sensor could not be determined.) The Supreme Court specifically overturns Federal Circuit precedent stating that, a claim is indefinite “only when it is 'not amenable to construction' or 'insolubly ambiguous'” 1). Instead, the Supreme Court holds that § 112 ¶2 requires:
“that a patent's claims, viewed in light of the specification and prosecution history, inform those skilled in the art about the scope of the invention with reasonable certainty. The definiteness requirement, so understood, mandates clarity, while recognizing that absolute precision is unattainable. The standard we adopt accords with opinions of this Court stating that “the certainty which the law requires in patents is not greater than is reasonable, having regard for their subject matter.” [Citations omitted].”
Remand from Supreme Court
On remand from the Supreme Court, in a separate opinion, the Federal Circuit reviewed the Supreme Court's decision, prior case law related to “reasonable certainty,” and the factual record relied upon in their initial decision to find that Biosig's claims satisfy 112 ¶2, and again held that the claims are not indefinite.
- Federal Circuit's Oral Arguments on Remand recorded January 7, 2015.
A petition for en banc review was later denied.
- Discussion and highlights
En Banc CAFC reaffirm written description doctrine (03/28/10)
In Ariad Pharmaceuticals et al v. Eli Lilly the CAFC upheld a separate written description requirement. The patent at issue relates to a method for inhibiting an intracellular mechanism for expressing genes driving an immune response. Ariad and its co-plaintiffs sued Eli Lilly for infringement of claims 80, 95, 144, and 145 of the patent. A jury returned a verdict that the claims were valid (meeting the enablement and written description requirments) and infringed. Eli Lilly appealed the Federal Circuit, which reversed the jury's verdict of validity on grounds that the patent fails to set forth an adequate written description in a decision rendered in April 2009 2). The CAFC agreed to rehear en banc to answer the following two questions:
- Whether 35 U.S.C. §112, paragraph 1, contains a written description requirement separate from an enablement requirement?
- If a separate written description requirement is set forth in the statute, what is the scope and purpose of that requirement?
In answering the first question, the CAFC held that the plain meaning of the statute does not limit the written description requirement solely to requiring enablement. Stated the court: “If Congress had intended enablement to be the sole description requirement of § 112, first paragraph, the statute would have been written differently” and “Finally, a separate requirement to describe one’s invention is basic to patent law. Every patent must describe an invention. It is part of the quid pro quo of a patent. . . .” The CAFC cited the Supreme Court cases Schriber-Schroth 3), Gill v. Wells 4), and Festo 5).
With regard to the scope of the requirement, the court held that:
- “the test for sufficiency is whether the disclosure of the application relied upon reasonably conveyed to those skilled in the art that the inventor had posession of the claimed subject matter as of the filing date.”
- “the specification must describe an invention understandable to that skilled artisan and show that the invention actually invented the invention claimed.”
- “the written description requirement does not demand either examples or an actual reduction to practice”
- “actual 'posession' or reduction to practice outside oft he specification is not enough.”
With regard to the purpose of the requirement, the court held that:
- “requiring a written description of the invention plays a vital role in curtailing claims that do not require undue experimentation to make and use, and thus satisfy enablement, but that have not been invented, and thus cannot be described.”
- “The written description requirement also ensures that when a patent claims a genus by its function or result, the specification recites sufficient materials to accomplish that function–a problem that is particularly acute in the biological arts.
The court does not sympathize with the impact of the written description requirement on research institutions (such as co-plaintiff MIT):
- “Patents are not awarded for academic theories, no matter how groundbreaking or necessary to the later patentable inventions of others. '[A] patent is not a hunting license. It is not a reward for the search, but compensation for its successful conclusion.'”
- “The goal is to get the right balance, and the written description doctrine does so by giving the incentive to actual invention and not 'attempt[s] to preempt the future before it has arrived. […] As this court has repeatedly stated, the purpose of the written description requirement is to 'ensure that the scope of the right to exclude, as set forth in teh claims, does not overreach the scope of the inventor's contribution to the field of art as described int he patent specification.”
Claim scope narrowed by spec; invalidated by §112, ¶1 (03/16/09)
In ICU Medical v. Alaris Medical Systems, the Fed. Cir. affirmed the district court's narrow construction of the term, “spike” as being “an elongated structure having a pointed tip for piercing the seal, which tip may be sharp or slightly rounded” stating that “it is 'entirely proper to consider the functions of an invention in seeking to determine the meaning of particular claim language.'” 6)
§112, ¶1: In addition, the Fed. Cir. affirmed the district court's finding of invalidity under §112 ¶1 of broader “spikeless” claims. The Fed. Cir. cited LizardTech v. Earth Resource Mapping 7) and rejected ICU's contention that “a person . . . would recognize that the specification discloses a preslit (or precut) seal that would permit fluid transmission without the piercing of a spike.” Stated the court: “It is not enough that it would have been obvious . . . that a preslit trampoline seal could be used without a spike. ICU has failed to point to any disclosure in the patent specification that describes a spikeless valve with a preslit trampoline seal” (citation omitted).
Cross posted: Claim Construction
Fed. Cir. finds "fragile" vague (01/27/07)
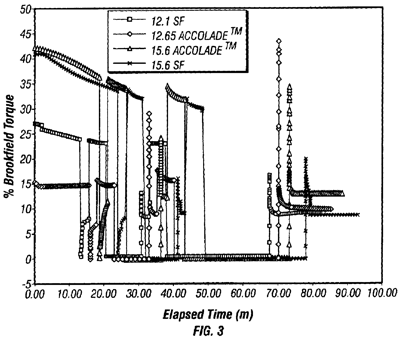 In Halliburton Energy Services, Inc. v. M-I LLC, the Fed. Cir. affirmed the district court's grant of summary judgment, agreeing that the term “fragile gel” is not amenable to construction or insolubly ambiguous and therefore indefinite (citing Datamize, LLC v. Plumtree Software, Inc. 8). The Fed. Cir. rejected Halliburton's arguments that a fragile gel is one that easily transitions from gel to liquid and back again is sufficiently objective so that a skilled artisan would understand the scope. Halliburton's suggestion that the “L-shaped” curve exhibited by a Brookfield viscometer shown in Figure 3 of the patent (reproduced here) identifies a fluid as being a “fragile gel” as rejected because, as Halliburton admits, the prior art 12.1 SF fluid also exhibits this feature. The Fed. Cir. also rejected Halliburton's arguments that fragile gel is defined as a gel that can suspend drill cuttings and weighting materials at rest because the quantity, weight, size and/or volume of cuttings to be suspended was undefined. The Fed. Cir. drew a parallel between this argument and that presented Geneva Pharmaceuticals, Inc. v. GlaxoSmithKline PLC, 9) regarding the phrase, “synergistically effective amount,” stating that in the present case, “an artisan would not know from one well to the next whether a certain drilling fluid was within the scope of the claims because a wide variety of factors could affect the adequacy.”
In Halliburton Energy Services, Inc. v. M-I LLC, the Fed. Cir. affirmed the district court's grant of summary judgment, agreeing that the term “fragile gel” is not amenable to construction or insolubly ambiguous and therefore indefinite (citing Datamize, LLC v. Plumtree Software, Inc. 8). The Fed. Cir. rejected Halliburton's arguments that a fragile gel is one that easily transitions from gel to liquid and back again is sufficiently objective so that a skilled artisan would understand the scope. Halliburton's suggestion that the “L-shaped” curve exhibited by a Brookfield viscometer shown in Figure 3 of the patent (reproduced here) identifies a fluid as being a “fragile gel” as rejected because, as Halliburton admits, the prior art 12.1 SF fluid also exhibits this feature. The Fed. Cir. also rejected Halliburton's arguments that fragile gel is defined as a gel that can suspend drill cuttings and weighting materials at rest because the quantity, weight, size and/or volume of cuttings to be suspended was undefined. The Fed. Cir. drew a parallel between this argument and that presented Geneva Pharmaceuticals, Inc. v. GlaxoSmithKline PLC, 9) regarding the phrase, “synergistically effective amount,” stating that in the present case, “an artisan would not know from one well to the next whether a certain drilling fluid was within the scope of the claims because a wide variety of factors could affect the adequacy.”
Intersection of §112 2¶2 validity and §102 anticipation: In response to Halliburton's arguments that a determination of whether the claim reads on the prior art 12.1 SF fluid should be reserved for a validity analysis under §102, the Fed. Cir. disagreed, holding that a evaluation of a claim's definiteness can include a determination as to whether the patent expressly or at least clearly differentiates itself from specific prior art. Stated the court: “Such differentiation is an important consideration in the definiteness inquiry because in attempting to define a claim term, a person of ordinary skill is likely to conclude that the definition does not encompass that which is expressly distinguished as prior art.”
Functional claim limitations: The Fed. Cir. gave the following advice when dealing with functional claim limitations:
When a claim limitation is defined in purely functional terms, the task of determining whether that limitation is sufficiently definite is a difficult one that is highly dependent on context (e.g., the disclosure in the specification and the knowledge of a person of ordinary skill in the relevant art area). We note that the patent drafter is in the best position to resolve the ambiguity in the patent claims, and it is highly desirable that patent examiners demand that applicants do so in appropriate circumstances so that the patent can be amended during prosecution rather than attempting to resolve the ambiguity in litigation.
A patent drafter could resolve the ambiguities of a functional limitation in a number of ways. For example, the ambiguity might be resolved by using a quantitative metric (e.g., numeric limitation as to a physical property) rather than a qualitative functional feature. The claim term might also be sufficiently definite if the specification provided a formula for calculating a property along with examples that meet the claim limitation and examples that do not. See Oakley, Inc. v. Sunglass Hut Int’l, 316 F.3d 1331, 1341 (Fed. Cir. 2003).
An additional blurb on this case is provided in the procedural matters section.
- Patent in suit: 6,887,832
Patent must enable "full scope of the claims" (03/22/07)
In Liebel-Flarsheim v. Medrad 10), the Fed. Cir. held that claims are not enabled even though a preferred embodiment is clearly enabled if the claims also read on non-enabled embodiments, in particular where the non-enabled embodiments requires a significant amount of experimentation to practice the non-enabled embodiment as evidenced by the specification teaching away from the non-enabled embodiments. The court distinguished Spectra-Physics noting that, while the court invalidated the claims in that case based on best mode but found a single disclosed attachment technique was sufficient to enable other attachment techniques within the scope of the claims, the other attachment techniques were well known in the art. With regard to a second claim asserted by Liebel, the court found the broad interpretation asserted by Liebel, which the Fed. Cir., in an earlier decision agreed with, was anticipated by an earlier patent by Medrad.
- Analysis and commentary (cached copy)
Fed. Cir. favors PTO on §112 first paragraph rejection (07/08/07)
<box green right 50% | MPEP 2163.04 I B. (in part):> In rejecting a claim, the examiner must set forth express findings of fact which support the lack of written description conclusion. . . . These findings should . . . (B) Establish a prima facie case by providing reasons why a person skilled in the art . . . would not have recognized that the inventor was in possession of the invention as claimed. . . . A simple statement such as “Applicant has not pointed out where the new (or amended) claim is supported, nor does there appear to be a written description of the claim limitation ___ in the application as filed.” may be sufficient where the claim is a new or amended claim, the support for the limitation is not apparent, and applicant has not pointed out where the limitation is supported.* </box | *emphasis added>
In Hyatt v. Dudas, the Fed. Cir. reversed the lower court's ruling that the PTO failed to make out a prima facie rejection under 35 U.S.C. §112, first paragraph. During prosecution of a continuation application with a family lineage going back decades, Hyatt withdrew all pending claims and submitted 1,100 new claims directed to various combinations of disclosed elements. The examiner initially rejected a representative sample of the claims for failing the written description requirement because while each individual element was disclosed in that specification, nowhere did Hyatt specify the particular configurations or combinations of elements claimed. The Fed. Cir. reversed holding “that § 2163.04(I)(B) [of the MPEP] as written is a lawful formulation of the prima facie standard for a lack of written description rejection.”
Claim invalidated under 35 U.S.C. §112 ¶4 (08/03/06)
In Pfizer, Inc., et al. v. Ranbaxy Laboratories, Limited, et al., Pfizer asserts claim 6, reading, “The hemicalcium salt of the compound of claim 2.” The court noted that 35 U.S.C. §112 4 states:
“Subject to the following paragraph [concerning multiple dependent claims], a claim in dependent form shall contain a reference to a claim previously set forth and then specify a further limitation of the subject matter claimed. A claim in dependent form shall be construed to incorporate by reference all the limitations of the claim to which it refers.”
Stated the court: Ranbaxy correctly argues that claim 6 fails to “specify a further limitation of the subject matter” of the claim to which it refers because it is completely outside the scope of claim 2. We must therefore reverse the district court with respect to this issue and hold claim 6 invalid for failure to comply with § 112, 4.”



